Help Solve a Global Crisis Through an Online Puzzle
Since 2008, the Foldit game has engaged the public in solving puzzles for science. Now it's tackling crop contamination.
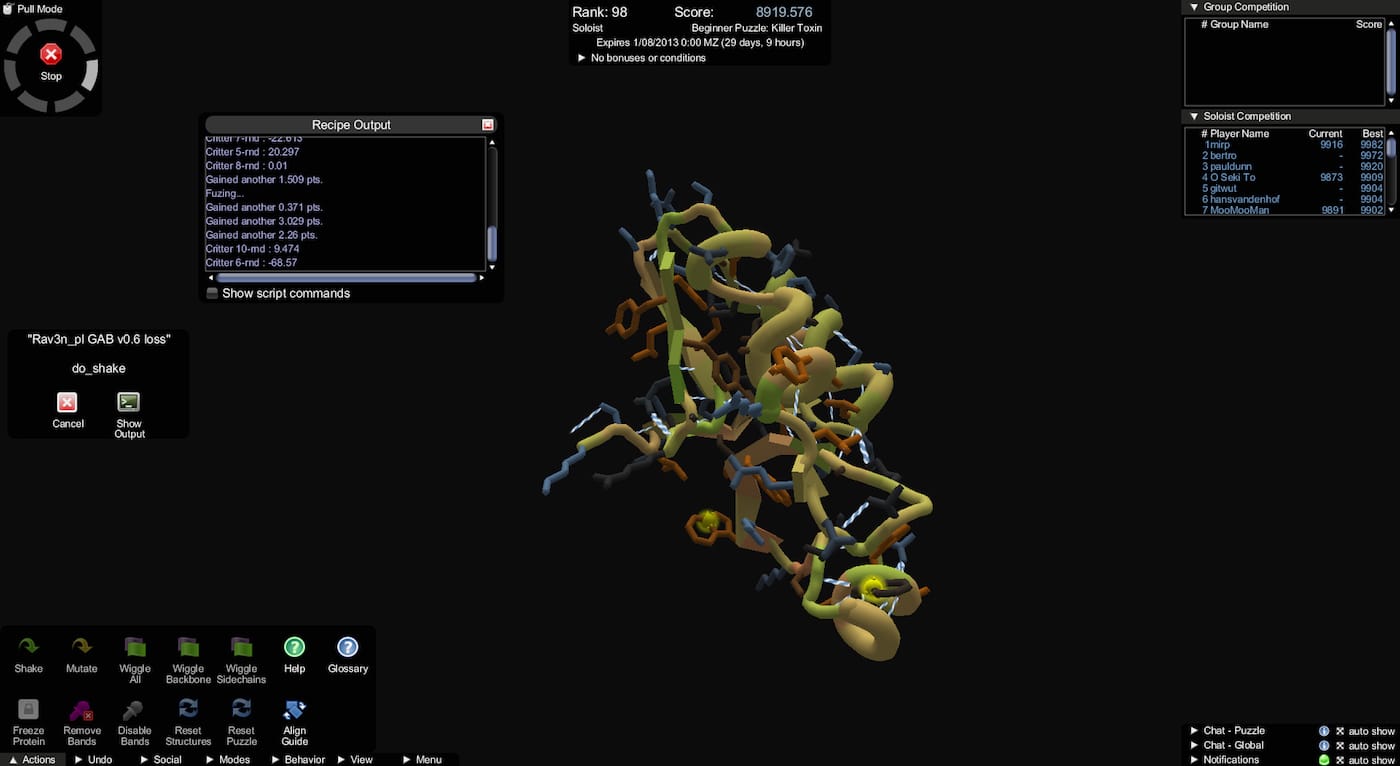
The University of Washington launched the online game Foldit in 2008, and since then it has successfully engaged the public in solving puzzles for science. The crowdsourcing initiative asks users to fold proteins into new and optimal three-dimensional configurations. This April, a New York Times article reported that it’s involved “nearly a million players over its lifetime,” including on puzzles about Ebola binding protein and the Marburg virus. In 2011, they helped decipher in a matter of weeks the structure of a protein-sniping enzyme that is critical in how the AIDS virus reproduces, and in 2012, it was reported that the players were the first to crowdsource a redesigned protein.
Now, Foldit is tackling crop contamination. On October 16, UC Davis announced a partnership between their university, Mars, Inc., and other partners to address aflatoxins through the University of Washington’s game. Aflatoxins are poisonous carcinogens produced by crop mold, and are a major concern for public health. UC Davis states that they contaminate “approximately a quarter of the food crops in the world,” and cause stunting when consumed by children, as well as around 90,000 cases of live cancer annually. Significantly, climate change could worsen this crisis.
In a report by Scientific American on the new puzzle, Justin Siegel, assistant professor of chemistry, biochemistry and molecular medicine at UC Davis, described Foldit as “three-dimensional Tetris,” but emphasized that it’s driven “by a physical reality of how stable this protein is.” Called “Aflatoxin Challenge: Round 1,” the puzzle asks players to begin with an enzyme that contains the potential for degrading aflatoxin’s toxicity, and then to improve it to neutralize the carcinogens.
Players who are successful at the challenge, which runs through October 31, will be tested in Siegel’s laboratory. The top designs will then be synthesized, and released into the public domain to contribute to global food safety. Foldit participants don’t have to be scientists, they just need a curious brain and a willingness to apply it to reworking these protein structures.
Aflatoxin Challenge: Round 1 is available to play on Foldit through October 31.





